Why "CTA" as a word is written?
• Functional neurosurgery
• Tumor ablation
• Biopsy procedures
• Catheter-based cardiovascular interventions
There was no unified, procedure-level visibility into:
• How many addressable procedures were performed annually?
• Which states had the highest MRI installed base?
• Which hospitals were already adopting intraoperative MRI (iMRI)?
• What percentage of procedures could realistically shift to MRI-guided workflows?
• What revenue per procedure could be expected?
The German company faced a classic commercialization dilemma:
“We know the technology is strong but where is the highest-converting market entry point?”
The Market Reality: A Massive but Unmapped Opportunity
BIS Research’s preliminary analysis revealed:
• 15 million U.S. adults suffer from knee osteoarthritis
• 18 million are affected by coronary artery disease
• Over 2.2 million relevant procedures are performed annually
More importantly:
• A 30% procedural shift toward MRI-guided approaches represented 675,000+ procedures
• Revenue potential exceeded $1.4 billion
• Revenue per procedure reached $2,090+
• 1,000+ hospitals were identified as viable targets
At the same time:
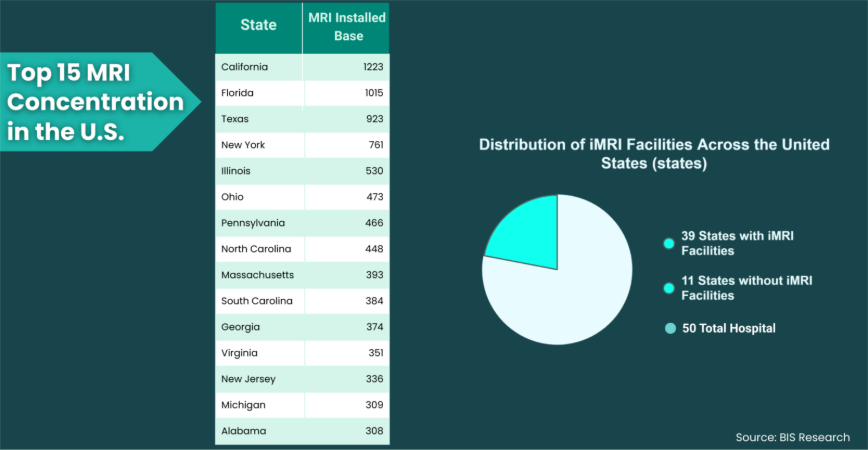
• iMRI facilities were already present in 39 U.S. states
• The top 15 states accounted for nearly 66% of MRI installations
• Low-field and helium-free MRI systems were driving installed base growth
The opportunity wasn’t speculative.
It was measurable.
But only with the right intelligence.
How BIS Research Helped: Turning Data into a Go-To-Market Blueprint
Instead of delivering a static market report, BIS Research deployed its Surgical Procedure Volume Database (SPVD) to build a structured commercialization roadmap.
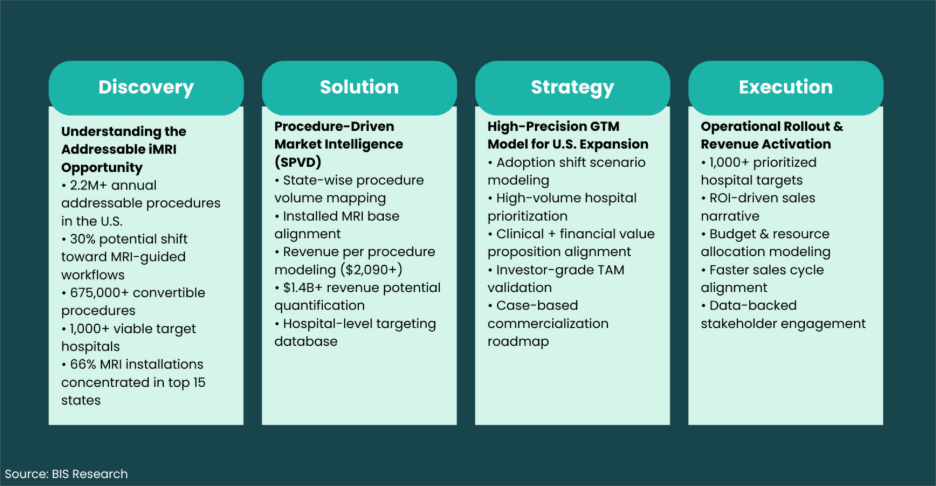
Step 1: Discovery
We mapped unmet needs and procedure gaps across key U.S. states.
Step 2: Opportunity Modeling
We quantified:
• Addressable procedure volumes
• Adoption shift scenarios
• Revenue per procedure
• State-wise concentration
• Installed base alignment
Step 3: Target Hospital Identification
Using MRI installed base and interventional adoption data, we identified 1,000+ high-potential hospitals .
Step 4: Case-Based GTM Strategy
We aligned:
• Clinical value proposition
• Revenue expansion narrative
• Budget modeling
• Operational rollout plan
The result?
A fully data-backed U.S. expansion strategy, not guesswork.
Why Surgical Procedure Volume Intelligence Is the Missing Link in MedTech Expansion
Most medical device companies rely on:
• Top-down TAM estimates
• Physician interviews
• Generic reimbursement assumptions
• Limited distributor insights
But in today’s competitive healthcare environment, that is not enough.
To win, you need:
Procedure-level volume data
State-level opportunity breakdown
Adoption shift modeling
Revenue-per-procedure visibility
Hospital-level targeting precision
Competitive installed base mapping
That is exactly what the Surgical Procedure Volume Database (SPVD) delivers.
The Strategic Impact: From Market Entry Risk to Revenue Certainty
For the German medical device manufacturer, SPVD enabled:
• Focused targeting instead of nationwide scatter strategy
• Prioritized engagement with high-volume hospitals
• Clear ROI narrative for hospital administrators
• Data-driven investor discussions
• Faster sales cycle alignment
Instead of asking,
“Is there opportunity in the U.S.?”
They could confidently say,
“There are 675,000+ procedures ready for conversion, representing a $1.4B+ revenue opportunity.”
That changes conversations.
That accelerates decisions.
That drives adoption.
Why This Matters Now: The MRI-Guided Intervention Shift Is Accelerating
With:
• Radiation-free imaging
• Superior soft-tissue visualization
• Real-time intraoperative guidance
• Growing adoption of low-field and helium-free MRI systems
Hospitals are increasingly evaluating MRI-integrated procedural suites.
The companies that win will be those who:
• Know where procedure volumes are concentrated
• Understand which states are early adopters
• Identify hospitals with capacity and alignment
• Enter the market with precision
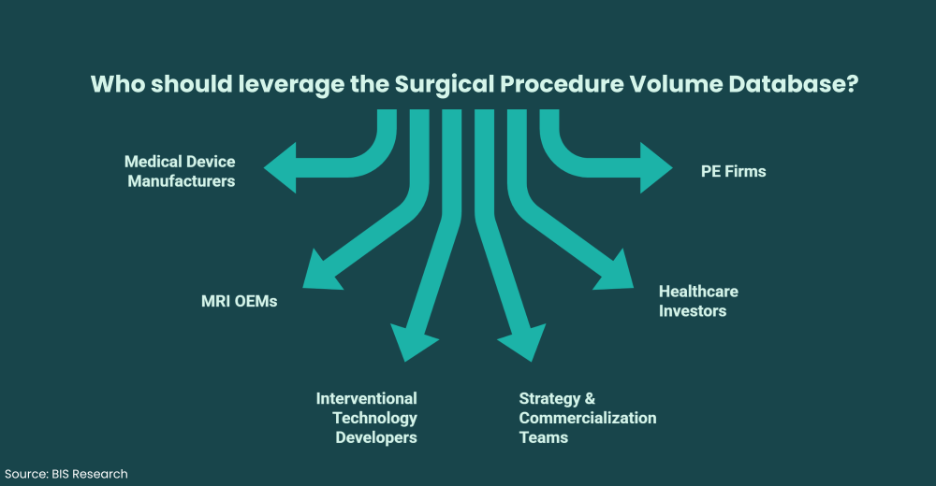
Who Should Leverage the Surgical Procedure Volume Database?
The SPVD is designed for:
• Medical device manufacturers planning U.S. expansion
• MRI OEMs targeting intraoperative applications
• Interventional technology developers
• Strategy & commercialization teams
• Healthcare investors conducting due diligence
• PE firms evaluating platform scalability
If your organization is asking:
• “Which hospitals should we target?”
• “What is the real addressable procedure market?”
• “What is the revenue potential by indication?”
• “Where will adoption accelerate first?”
Then surgical procedure volume intelligence is not optional it is essential.
Move from Assumptions to Actionable Intelligence
Entering the U.S. without granular surgical data is like navigating without a map.
The German medical device company chose clarity.
They chose structured, defensible data.
They chose SPVD.
Now it’s your move.
Ready to Identify High-Volume Hospitals and Quantify Your Revenue Opportunity?
If you are planning:
• A U.S. market/global entry strategy
• A product launch in MRI-guided interventions
• A hospital targeting campaign
• An investor-grade opportunity assessment
Explore the Surgical Procedure Volume Database by BIS Research
Identify 1,000+ target hospitals
Quantify procedure-driven revenue potential
Build a data-backed commercialization roadmap



