
Cell and gene therapies (CGTs) are reshaping modern medicine by addressing diseases at their genetic and cellular roots. However, the success of these therapies depends heavily on drug delivery technologies, systems that ensure genetic material or engineered cells reach the right target safely and efficiently. In 2025, drug delivery emerged as one of the most active innovation areas within the CGT ecosystem, driven by clinical progress, regulatory momentum, and manufacturing needs.
Unlike conventional pharmaceuticals, cell and gene therapies require precise intracellular delivery of DNA, RNA, or modified cells. Inefficient delivery can result in immune reactions, off-target effects, or therapeutic failure. As CGTs move from rare diseases toward broader indications such as oncology, neurology, and cardiology, delivery platforms must become safer, scalable, and repeatable.
In 2025, the industry’s focus shifted from proof-of-concept toward optimization, targeting, and manufacturability.
Explore the full market potential
schedule a consultation
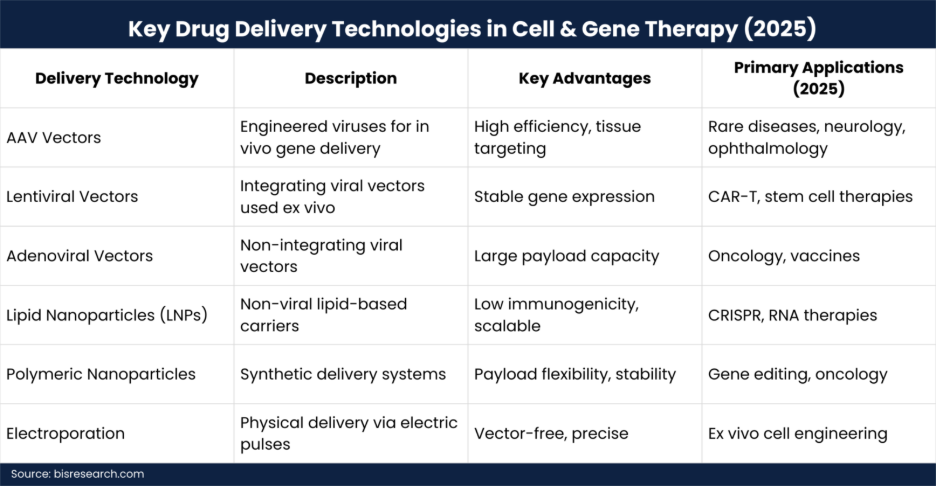
Viral Vectors: Still Dominant, Now More Refined
Viral vectors remain the backbone of cell and gene therapy drug delivery due to their high transduction efficiency.
Key viral platforms in 2025:
In 2025, major advancements included capsid engineering, tissue-specific targeting, and immune-evasion strategies. These innovations helped reduce dose requirements and improve safety, addressing long-standing concerns around immunogenicity and repeat dosing.
Want deeper insights?
[Download Sample Report Now]
While viral vectors dominate, non-viral delivery technologies gained significant traction in 2025, particularly for gene editing and RNA-based therapies.
Key non-viral technologies:
Non-viral systems are increasingly favored for their payload flexibility, lower cost potential, and suitability for repeated administration—a key requirement for chronic diseases.
One of the most important developments in 2025 was the emergence of precision delivery technologies:
These advances significantly expanded the therapeutic scope of CGTs into neurodegenerative diseases, solid tumors, and autoimmune disorders.
2025 marked a year of increased regulatory confidence in CGT platforms:
These trends reinforced the importance of robust, scalable drug delivery systems.
Several companies led innovation across delivery platforms:
Large pharmaceutical companies such as AstraZeneca and Eli Lilly also expanded CGT delivery capabilities through acquisitions and platform investments.
By the end of 2025, cell and gene therapy drug delivery had evolved from a technical hurdle into a strategic differentiator. The future lies in targeted, repeatable, and scalable delivery platforms that can support broader patient populations.
As delivery technologies continue to mature, they will play a decisive role in making curative therapies safer, more accessible, and commercially viable.
Looking to enter a new market but unsure where to start? At BIS Research, we provide first-hand insights directly from key opinion leaders (KOLs), backed by rigorous primary and secondary research.