
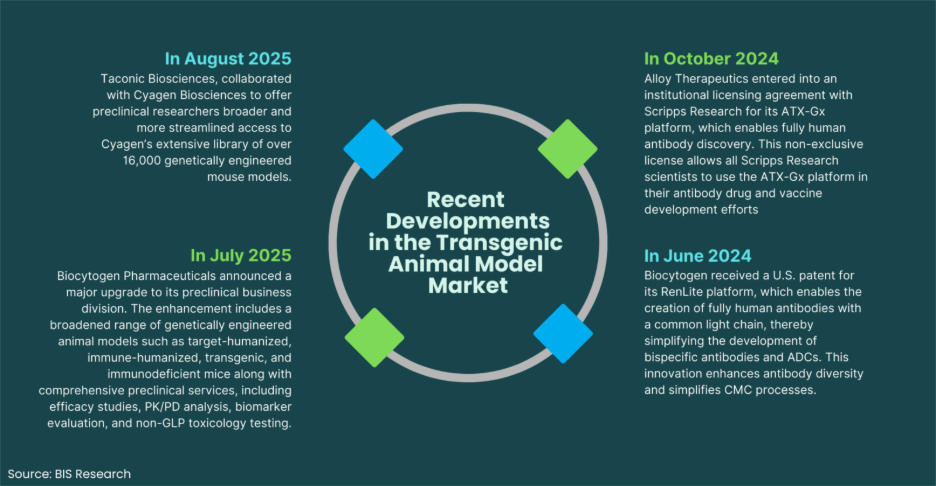
Transgenic animal models remain the backbone of modern biomedical research, powering breakthroughs in oncology, immunology, rare diseases, and regenerative medicine. As drug pipelines grow more complex and precision medicine becomes mainstream, the demand for highly accurate, genetically engineered animal models has intensified.
What has truly accelerated this field, however, is not just demand but technology. Over the past two decades, gene-editing platforms have evolved from labor-intensive and time-consuming methods to highly precise, scalable systems capable of modeling human disease with unprecedented accuracy.
Here are five breakthrough technologies transforming transgenic animal research today.
CRISPR/Cas9 has fundamentally reshaped the transgenic animal landscape. Its ability to introduce targeted genetic modifications quickly, efficiently, and cost-effectively has made it the dominant genome-editing tool globally.
Unlike earlier technologies, CRISPR relies on a guide RNA to direct the Cas9 enzyme to a specific DNA sequence, enabling precise gene knockouts, insertions, or modifications. The simplicity of design and multiplexing capability allows researchers to edit multiple genes simultaneously critical for modeling complex, polygenic diseases such as cancer and autoimmune disorders.
For pharmaceutical R&D teams, CRISPR significantly shortens model development timelines, accelerating preclinical validation and target discovery. It has also enabled the development of “humanized” animal models that express human immune components, making them indispensable in immuno-oncology and biologics testing.
Request a sample report on Transgenic animal models Market
Before CRISPR’s rise, Transcription Activator-Like Effector Nucleases (TALENs) were considered a major leap forward in genome engineering. TALENs are engineered proteins that bind to specific DNA sequences and induce double-strand breaks, enabling targeted modifications.
Although more labor-intensive to design compared to CRISPR, TALENs offer exceptional specificity and lower off-target activity in certain contexts. This makes them valuable for applications requiring highly precise edits or when CRISPR’s PAM sequence requirements limit accessibility.
TALENs have been particularly useful in generating transgenic large-animal models, such as pigs and cattle, where precision is essential for translational research and xenotransplantation studies.
Zinc Finger Nucleases (ZFNs) represent one of the earliest targeted genome-editing technologies. They combine zinc finger DNA-binding domains with a nuclease enzyme to create site-specific double-strand breaks.
While ZFNs are more complex to engineer and comparatively expensive, they laid the groundwork for modern gene-editing strategies. Their legacy persists in certain proprietary and clinical research settings, where validated ZFN platforms remain in use.
Importantly, ZFNs helped establish proof-of-concept for targeted genome engineering, paving the way for TALENs and CRISPR to scale the field further. In transgenic animal research, they enabled early knockout and knock-in models that expanded disease modeling capabilities significantly.
Long before programmable nucleases emerged, embryonic stem (ES) cell microinjection was the gold standard for generating transgenic animals particularly mice.
This technique involves genetically modifying embryonic stem cells in vitro and then injecting them into blastocysts, which are implanted into surrogate mothers. The resulting offspring carry the desired genetic modification.
Though time-consuming and technically demanding, ES cell microinjection remains valuable for complex genetic constructs and conditional alleles. Many foundational disease models in oncology and developmental biology were developed using this approach.
Even today, it complements CRISPR-based strategies, especially when precise control over insertion sites is required.
Download the complete TOC now!
Conditional knockout systems, such as Cre-loxP and Flp-FRT recombination, allow genes to be deactivated in specific tissues or at specific developmental stages. This is crucial when studying genes that are embryonically lethal if fully knocked out.
By enabling spatial and temporal gene control, conditional knockout technologies provide researchers with refined disease models that more accurately mimic human pathology. For example, tissue-specific gene silencing in liver or neural tissue has transformed metabolic and neurodegenerative disease research.
When combined with CRISPR editing, conditional systems offer unparalleled flexibility in designing next-generation transgenic models.
Book a Preview on Transgenic Animal Model Market
Together, these five technologies have reshaped transgenic animal research from a slow, niche capability into a scalable, high-precision industry supporting global pharmaceutical innovation. As biologics, gene therapies, and personalized medicine continue to expand, demand for increasingly sophisticated animal models will grow.
The next frontier will likely involve integrating gene-editing tools with AI-driven model design and advanced phenotyping platforms further enhancing translational accuracy while addressing ethical and efficiency concerns.
For biotech innovators, mastering these technologies is no longer optional it is strategic.